Anti-EBV Latent Membrane Protein 1 [CS 1-4]
Invented by Prof Martin Rowe from University Of Birmingham
Invented at University Of Birmingham
- Datasheet
- References (17)
- Inventor Info
Info
| Catalogue Number | 151471 |
| Applications | IHC IF IP WB |
| Antigen/Gene or Protein Targets | Epstein-Barr Virus, Latent Membrane Protein 1 (EBV-LMP1) |
| Reactivity | Virus |
| Relevance |
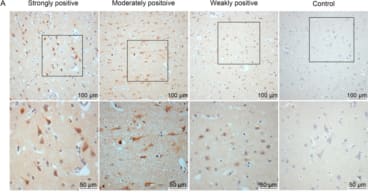
Anti-EBV (CS1) is a latent membrane protein 1 antibody, which detects a specific epitope upon LMP fusion protein in B-cell transformations, following EBV infection. Background and Research Application A combination of four pooled antibodies which collectively detect the latent membrane protein (LMP) of EBV, an important effector protein in B-cell transformation under EBV infection, across 20 geographically distinct EBV isolates. EBV is a human herpesvirus that establishes a life-long persistence in the host. The virus infects the vast majority of the world's adult population and is well known for its association with a broad spectrum of benign and malignant diseases. These include infectious mononucleosis, Burkitt's lymphoma, nasopharyngeal carcinoma, and is causally associated with lymphoid and epithelial malignancies, including post-transplant lymphoproliferative disorders, Hodgkin's disease, anaplastic nasopharyngeal carcinoma and gastric carcinomas. Latent membrane protein 1 (LMP1) of Epstein-Barr virus (EBV) is a transforming protein that affects multiple cell signalling pathways and contributes to EBV-associated oncogenesis. LMP1 can be expressed in some states of EBV latency, and significant induction of full-length LMP1 is also observed frequently during virus reactivation into the lytic cycle. LMP1 is critical for EBV-infected cell activation, adhesion and survival, and is usually expressed in the malignant cells. These antibodies were created to examine various aspects of LMP expression in B-cell lines transformed in vitro, detecting LMPs from 20 geographically varied EBV isolates. |
| Host | Mouse |
| Immunogen UniProt ID | P03230 |
| Positive Control | EBV transformed lymphoblastoid cell lines |
| Subclass | IgG1 kappa |
| Myeloma Used | P3X63Ag8.653 |
| Notes |
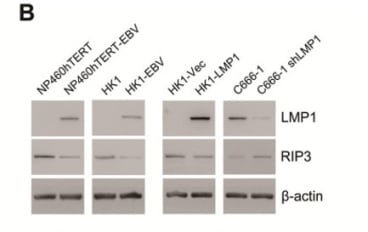
Production Details Purified using multi-step affinity chromatography with protein A. Storage Conditions Store at -20 degrees frozen. Avoid repeated freeze/thaw cycles. Points of Interest Anti-EBV LMP 1, along with 2-4, showed the expression of full length LMP was strongly inducible by phorbol ester and/or sodium butyrate not just in cells entering a lytic cycle. This indicates expression of LMP in EBV transformed B-cells is under specific regulations. Concentration 1mg/ml as standard Difference Between Clones 4 antibodies directed against EBV LMP1 were produced in the original experiment, CS1-4. Anti-EBV (CS1) reacted strongly with both of the major pKH548 bands, whilst CS2-4 only reacted with the higher 150kDa band. This suggests CS1 recognises an epitope within the LMP fusion protein distinct to the other three anti-LMP antibodies. All antibodies are specific for the carboxy terminus of LMP 1 aa 186–386 |
| Research Area | Adhesion, Cancer, Cell Signaling & Signal Transduction, Immunology, Virology |
References: 17 entries
Ye et al. 2020. J Cancer. 11(5):1257-1269. PMID: 31956372.
WB
Shi et al. 2019. Theranostics. 9(9):2424-2438. PMID: 31131045.
Wang et al. 2017. Virol Sin. 32(5):423-430. PMID: 29116594.
IHC
Martin et al. 2016. J Virol. 90(19):8520-30. PMID: 27440880.
WB
Castrale et al. 2011. J Transplant. 2011:865957. PMID: 21559262.
Flanagan et al. 2003. J Gen Virol. 84(Pt 7):1871-9. PMID: 12810882.
Localization of the Epstein-Barr virus protein LMP 1 to exosomes.
Europe PMC ID: 12810882
Nakatsuka et al. 2002. J Clin Oncol. 20(20):4255-60. PMID: 12377970.
Pyothorax-associated lymphoma: a review of 106 cases.
Europe PMC ID: 12377970
Xu et al. 2002. J Virol. 76(8):4080-6. PMID: 11907247.
Preferential localization of the Epstein-Barr virus (EBV) oncoprotein LMP-1 to nuclei in human T cells: implications for its role in the development of EBV genome-positive T-cell lymphomas.
Europe PMC ID: 11907247
Ascani et al. 1997. Ann Oncol. 8(11):1133-8. PMID: 9426333.
Pyothorax-associated lymphoma: description of the first two cases detected in Italy.
Europe PMC ID: 9426333
Epstein-Barr virus (EBV)-associated lymphoproliferative disease in the SCID mouse model: implications for the pathogenesis of EBV-positive lymphomas in man.
Europe PMC ID: 1845872
Rowe et al. 1991. J Exp Med. 173(1):147-58. PMID: 1845872.
IF
Rowe et al. 1987. J Gen Virol. 68 ( Pt 6):1575-86. PMID: 2438376.
Monoclonal antibodies to the latent membrane protein of Epstein-Barr virus reveal heterogeneity of the protein and inducible expression in virus-transformed cells.
Europe PMC ID: 2438376
Add a reference
References: 17 entries
Ye et al. 2020. J Cancer. 11(5):1257-1269. PMID: 31956372.
WB
Shi et al. 2019. Theranostics. 9(9):2424-2438. PMID: 31131045.
Wang et al. 2017. Virol Sin. 32(5):423-430. PMID: 29116594.
IHC
Martin et al. 2016. J Virol. 90(19):8520-30. PMID: 27440880.
WB
Castrale et al. 2011. J Transplant. 2011:865957. PMID: 21559262.
Flanagan et al. 2003. J Gen Virol. 84(Pt 7):1871-9. PMID: 12810882.
Localization of the Epstein-Barr virus protein LMP 1 to exosomes.
Nakatsuka et al. 2002. J Clin Oncol. 20(20):4255-60. PMID: 12377970.
Pyothorax-associated lymphoma: a review of 106 cases.
Xu et al. 2002. J Virol. 76(8):4080-6. PMID: 11907247.
Preferential localization of the Epstein-Barr virus (EBV) oncoprotein LMP-1 to nuclei in human T cells: implications for its role in the development of EBV genome-positive T-cell lymphomas.
Ascani et al. 1997. Ann Oncol. 8(11):1133-8. PMID: 9426333.
Pyothorax-associated lymphoma: description of the first two cases detected in Italy.
Epstein-Barr virus (EBV)-associated lymphoproliferative disease in the SCID mouse model: implications for the pathogenesis of EBV-positive lymphomas in man.
Rowe et al. 1991. J Exp Med. 173(1):147-58. PMID: 1845872.
IF
Rowe et al. 1987. J Gen Virol. 68 ( Pt 6):1575-86. PMID: 2438376.
Monoclonal antibodies to the latent membrane protein of Epstein-Barr virus reveal heterogeneity of the protein and inducible expression in virus-transformed cells.
Add a reference



![Image thumbnail for Anti-EBV Latent Membrane Protein 1 [CS 1-4]](https://res.cloudinary.com/ximbio/image/upload/c_fit,fl_lossy,q_auto/079341e3-36b2-4825-9804-4d3e7226b8bb.jpg)
![Image thumbnail for Anti-EBV Latent Membrane Protein 1 [CS 1-4]](https://res.cloudinary.com/ximbio/image/upload/c_fit,fl_lossy,q_auto/fb6a365f-e24a-440c-a498-721c7a8bfc02.jpg)
![Image thumbnail for Anti-EBV Latent Membrane Protein 1 [CS 1-4]](https://res.cloudinary.com/ximbio/image/upload/c_fit,fl_lossy,q_auto/d302965a-ef6c-46f3-b44b-11e0ae848b38.jpg)
![Image thumbnail for Anti-EBV Latent Membrane Protein 1 [CS 1-4]](https://res.cloudinary.com/ximbio/image/upload/c_fit,fl_lossy,h_45,q_auto/079341e3-36b2-4825-9804-4d3e7226b8bb.jpg)
![Image thumbnail for Anti-EBV Latent Membrane Protein 1 [CS 1-4]](https://res.cloudinary.com/ximbio/image/upload/c_fit,fl_lossy,h_45,q_auto/fb6a365f-e24a-440c-a498-721c7a8bfc02.jpg)
![Image thumbnail for Anti-EBV Latent Membrane Protein 1 [CS 1-4]](https://res.cloudinary.com/ximbio/image/upload/c_fit,fl_lossy,h_45,q_auto/d302965a-ef6c-46f3-b44b-11e0ae848b38.jpg)


