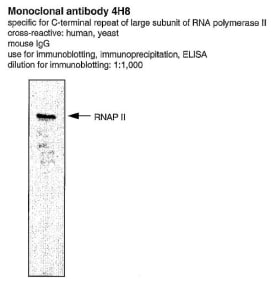
Anti-RNA polII [CTD 4H8]
Invented by Dr Jesper Svejstrup from Francis Crick Institute
Invented at Cancer Research UK London Research Institute: Clare Hall Laboratories
- Datasheet
- References (20)
- Inventor Info
Info
| Catalogue Number | 151247 |
| Applications | ChIP ELISA FACS IHC IF IP WB ChIP-seq |
| Antigen/Gene or Protein Targets | RNA Polymerase II |
| Reactivity | Human and Saccharomyces cerevisiae |
| Relevance | RNAPII is an essential component of the RNAP II transcription elongation complex. Recruitment of RNA processing factors to the elongation complex is coordinated by the phosphorylation of Serine-2 and Serine-5 residues within the C-terminal repeat (YSPTSPS) domain of the largest subunit of RNAPII. Mutations that directly affect transcription by RNA polymerases rank among the most central mediators of malignant transformation. CTD4H8 can also be used in chromatin immunoprecipitation (ChIP) assays. |
| Host | Mouse |
| Immunogen | Peptide: 10 repeats of synthetic peptide YSPTSPS using chemically synthesized phospho-Ser5 |
| Positive Control | HeLa cell line. |
| Subclass | IgG1 |
| Myeloma Used | Sp2/0-Ag14 |
| Recommended Growing Conditions | DMEM + 5% FCS |
| Strain | Balb/c |
| Notes |
Please note that this antibody detects both unphosphorylated and phosphorylated forms of the protein. When using this antibody in western blot and IP, it recognizes both the II0 and IIA form of RPB1 (ie the largest RNAPII subunit). These different forms of RPB1 (as seen in gels) have traditionally been loosely defined as ‘hyper-‘ and ‘hypo-phosphorylated’, respectively. Here, however, it is important to remember that RPB1 contains no less than 52 repeats of a heptapeptide (TSPTSPS) that can be phosphorylated at different positions (Tyr1, Ser2, Thr4, Ser5 and Ser7; this antibody was raised against a synthetic peptide with 10 of these repeats, mostly but probably not entirely, chemically phosphorylated at Ser5). So, when we say that it recognizes both forms (II0/hyper-phos and IIA/hypo-phos), it might really just mean that the antibody recognizes a few phosphorylated Ser5s among 52 otherwise mostly un-phosphorylated repeats. Alternatively, it might conceivably (though less likely) recognize UN-phosphorylated repeats as there might conceivably be a few of those even in the II0/hyper-phos RPB1 form. |
| Research Area | Epigenetics & Nuclear Signalling |
References: 20 entries
Williamson et al. 2017. Cell. 168(5):843-855.e13. PMID: 28215706.
WB
UV Irradiation Induces a Non-coding RNA that Functionally Opposes the Protein Encoded by the Same Gene.
Europe PMC ID: 28215706
Pang et al. 2014. Oncogene. 33(31):4039-49. PMID: 24141769.
ChIP
Seo et al. 2014. Nucleic Acids Res. 42(2):1016-25. PMID: 24163101.
Genome-wide reorganization of histone H2AX toward particular fragile sites on cell activation.
Europe PMC ID: 24163101
A functional interaction of E7 with B-Myb-MuvB complex promotes acute cooperative transcriptional activation of both S- and M-phase genes. (129 c).
Europe PMC ID: 24141769
Keren-Shaul et al. 2013. PLoS One. 8(1):e53506. PMID: 23326444.
Pre-mRNA splicing is a determinant of nucleosome organization.
Europe PMC ID: 23326444
Harrigan et al. 2011. J Cell Biol. 193(1):97-108. PMID: 21444690.
Replication stress induces 53BP1-containing OPT domains in G1 cells.
Europe PMC ID: 21444690
Li et al. 2006. Mol Biol Cell. 17(9):3848-59. PMID: 16807355.
Sir2 represses endogenous polymerase II transcription units in the ribosomal DNA nontranscribed spacer.
Europe PMC ID: 16807355
Yart et al. 2005. Mol Cell Biol. 25(12):5052-60. PMID: 15923622.
The HRPT2 tumor suppressor gene product parafibromin associates with human PAF1 and RNA polymerase II.
Europe PMC ID: 15923622
Engelhardt et al. 2005. J Virol. 79(9):5812-8. PMID: 15827195.
Association of the influenza A virus RNA-dependent RNA polymerase with cellular RNA polymerase II.
Europe PMC ID: 15827195
Kristjuhan et al. 2003. Proc Natl Acad Sci U S A. 100(13):7551-6. PMID: 12796514.
Spreading of Sir3 protein in cells with severe histone H3 hypoacetylation.
Europe PMC ID: 12796514
Kristjuhan et al. 2002. Mol Cell. 10(4):925-33. PMID: 12419235.
Transcriptional inhibition of genes with severe histone h3 hypoacetylation in the coding region.
Europe PMC ID: 12419235
Add a reference
References: 20 entries
Williamson et al. 2017. Cell. 168(5):843-855.e13. PMID: 28215706.
WB
UV Irradiation Induces a Non-coding RNA that Functionally Opposes the Protein Encoded by the Same Gene.
Pang et al. 2014. Oncogene. 33(31):4039-49. PMID: 24141769.
ChIP
Seo et al. 2014. Nucleic Acids Res. 42(2):1016-25. PMID: 24163101.
Genome-wide reorganization of histone H2AX toward particular fragile sites on cell activation.
A functional interaction of E7 with B-Myb-MuvB complex promotes acute cooperative transcriptional activation of both S- and M-phase genes. (129 c).
Keren-Shaul et al. 2013. PLoS One. 8(1):e53506. PMID: 23326444.
Pre-mRNA splicing is a determinant of nucleosome organization.
Harrigan et al. 2011. J Cell Biol. 193(1):97-108. PMID: 21444690.
Replication stress induces 53BP1-containing OPT domains in G1 cells.
Li et al. 2006. Mol Biol Cell. 17(9):3848-59. PMID: 16807355.
Sir2 represses endogenous polymerase II transcription units in the ribosomal DNA nontranscribed spacer.
Yart et al. 2005. Mol Cell Biol. 25(12):5052-60. PMID: 15923622.
The HRPT2 tumor suppressor gene product parafibromin associates with human PAF1 and RNA polymerase II.
Engelhardt et al. 2005. J Virol. 79(9):5812-8. PMID: 15827195.
Association of the influenza A virus RNA-dependent RNA polymerase with cellular RNA polymerase II.
Kristjuhan et al. 2003. Proc Natl Acad Sci U S A. 100(13):7551-6. PMID: 12796514.
Spreading of Sir3 protein in cells with severe histone H3 hypoacetylation.
Kristjuhan et al. 2002. Mol Cell. 10(4):925-33. PMID: 12419235.
Transcriptional inhibition of genes with severe histone h3 hypoacetylation in the coding region.
Add a reference


![Image thumbnail for Anti-RNA polII [CTD 4H8]](https://res.cloudinary.com/ximbio/image/upload/c_fit,fl_lossy,q_auto/7f8bd53e-0be3-4590-99f8-21ee55b63106.jpg)
![Image thumbnail for Anti-RNA polII [CTD 4H8]](https://res.cloudinary.com/ximbio/image/upload/c_fit,fl_lossy,h_45,q_auto/7f8bd53e-0be3-4590-99f8-21ee55b63106.jpg)


