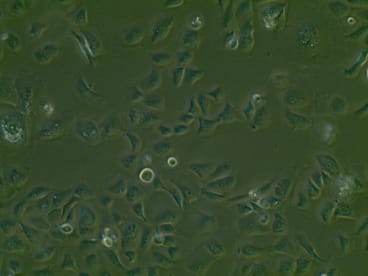
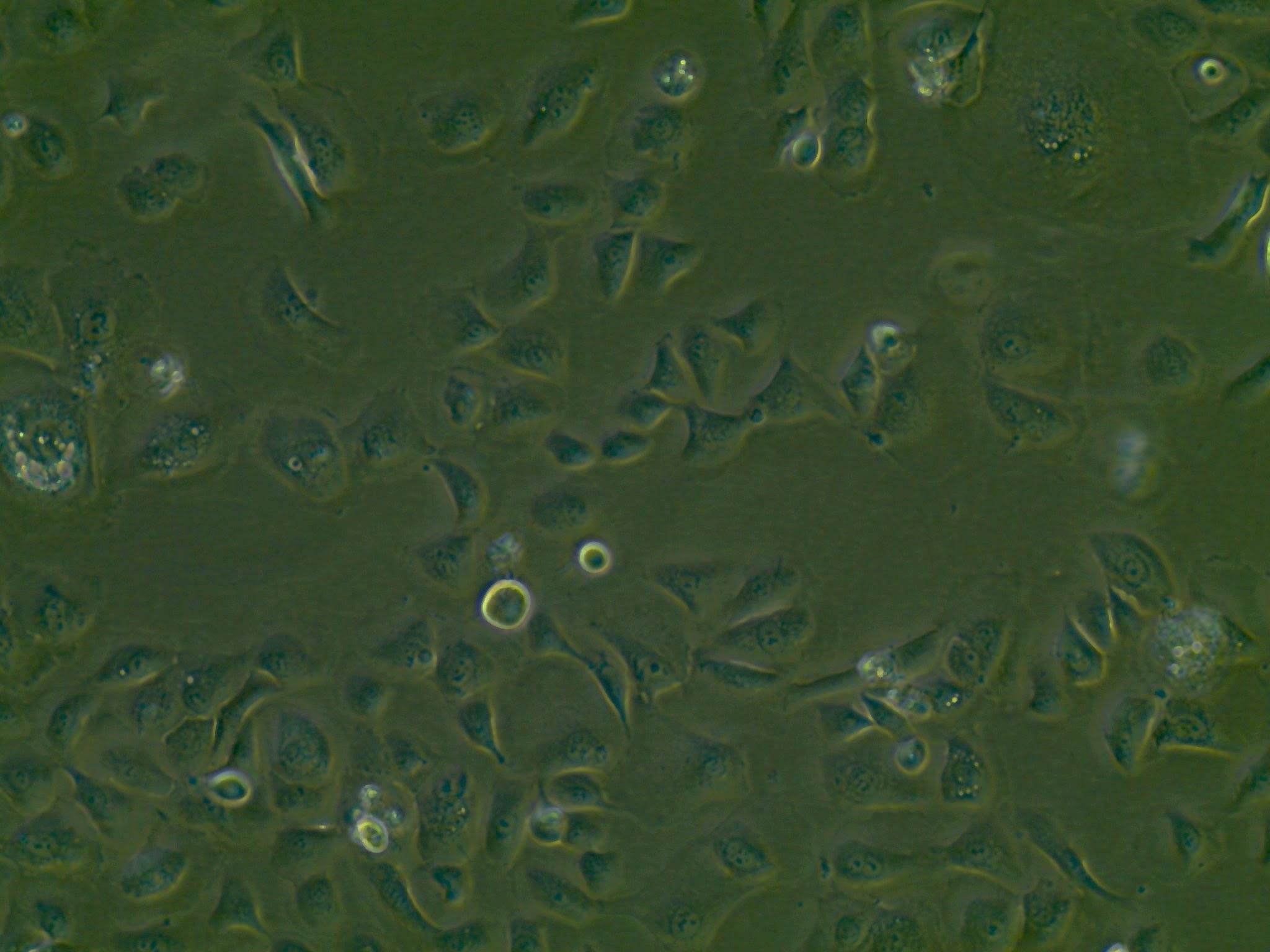
SG01 Cell Line
Invented by Jason Powell from University of Newcastle upon Tyne , Dr Chris Ward from University of Newcastle upon Tyne
Invented at University of Newcastle upon Tyne
- Datasheet
- References (2)
- Inventor Info
Info
| Catalogue Number | 153460 |
| Parental Line | Subglottic brushing specimen |
| Host | Human |
| Tissue | Subglottis |
| Disease Keywords | Subglottic stenosis, Cancer, Inflammation |
| Model | Immortalised Line |
| Relevance | The subglottic airway is an important gatekeeper that functions to prevent lower lung infection and is a crucial area in terms of inflammation (subglottic stenosis) and malignancy. |
| Production Details | The SG01 Cell Line was derived from a single patients neoplasia-free human subglottic brushing specimen. The cells were immortalised using a SV40 Cell immortalization system, utilizing a recombinant lentiviral vector. SG01 were successfully lifted onto an air-liquid interface (ALI) culture system, using the appropriate growth medium. At ALI cells demonstrated cilia coverage and mucus production. Cells demonstrate tight epithelial junctions and relevant ion channels expression, including ENaC, CFTR and calcium-activated chloride channels. |
| Conditional | No |
| Research Area | Cancer, Drug Discovery & Development |
| Growth/Phenotype Keywords | Adherent |
| Recommended Growing Conditions | SG01 cells were cultured on a collagen coated container in Bronchial Epithelial Cell Growth Medium (BEGM) (available from Clonetics Corporation, CC3171 (BEBM) plus additives CC4175 or BEGM Bullet Kit, CC3170). , supplemented with 100 µg/mL streptomycin and 100 U/mL penicillin at 37°C and 5% CO2. Split sub-confluent cultures (70-80%) 1:3 to 1:6 using 0.05% trypsin/EDTA. |
| Notes |
How to plate the cells from frozen ampule: Prepare a flask coated with 10ug/cm2 Collagen (Type 1) at the following volumes 2.5mls/25cm2; 7.5ml/75cm2 and 17.25mls/175cm2. (Sigma 0.1% solution, 8919-20ML), diluted 1:10 in growth medium (BEGM). Add mixture at ratio of 0.1 ml per cm² surface area. Incubate at 37°C for approximately 6 hours and 5% CO2, or overnight at 4°C. Remove excess fluid prior to use and wash flask with either PBS or media. Seeding density at first seed and subculture: Resuscitation: A centrifugation step (100-150g for 2-3 minutes to pellet cells) to remove the cryoprotectant is essential. Rapidly thaw the frozen ampoule in a water bath at 37°C for 1-2 minutes. Transfer the contents to a centrifuge tube and slowly add 5-10ml of pre-warmed growth media. Remove a sample for counting. Centrifuge at 100-150g for 2-3 minutes to pellet cells and seed at ~10, 000 cells per cm². Seed cells thereafter into new flasks between 3,000 and 10, 000 cells per cm². Passage reagents/tools: Check cultures daily and media change as required. Subculture sub-confluent cultures (maximum 70% confluence) using 0.05% trypsin/EDTA. Incubate for 5-10 min until cells detach. Note: BEGM medium is almost serum-free, therefore use of a trypsin inhibitor is essential. Add an equal or greater volume of used growth medium supplemented with 10% FBS or Trypsin Inhibitor (Soybean) to stop digestion. Centrifuge at 200-250g for 5 minutes. The supernatant is removed and the cell pellet re-suspended with fresh BEGM medium prior to transfer to another growth flask. Cryopreservation medium: Harvest the cells at 70-80% confluency using 0.05% trypsin/EDTA, using growth medium supplemented with 10% FBS as described previously. When the cells are dislodged, they are centrifuged at 200g at 10°C for 7 minutes. The supernatant is discarded and the pellet resuspended in freeze media (45% Conditioned medium, 45% Fresh medium and 10% Dimethyl sulfoxide (DMSO)). |
References: 2 entries
Establishment of an immortalized human subglottic epithelial cell line
A Human in Vitro Model of the Subglottic Airway
Add a reference
References: 2 entries
Establishment of an immortalized human subglottic epithelial cell line
A Human in Vitro Model of the Subglottic Airway
Add a reference