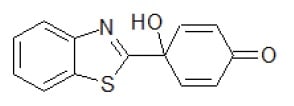
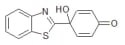
Antitumoral Quinol2 Small Molecule (Tool Compound)
Invented at University of Nottingham
- Datasheet
- References (6)
- Inventor Info
Info
| Catalogue Number | 151840 |
| Antigen/Gene or Protein Targets | Thioredoxin reductase |
| Type | Inhibitor |
| Relevance | The antitumoral Quinol2, also known as PMX464, is a potent in vitro antitumour activity was observed in HCT 116 and HT 29 human colon as well in as MCF-7 and MDA 468 human breast cancer cell lines. This molecule also showed in vivo antitumour activity against human RXF 944XL renal xenografts in nude NMRI mice.A new class of molecule (4-hydroxy-4-hetarylcyclohexa-2,5-dien-1-ones) bearing a new pharmacophore (‘quinols’) has been identified with potent activity against renal, colon and breast cell lines in the NCI 60-cell panel. It also demonstrates potent in vitro anti-proliferative activity and in vivo anti-tumour activity in tumour xenografts |
| Molecular Formula | C13H9NO2S |
| lUPAC | 4-Benzothiazol-2-yl-4-hydroxycyclohexa-2,5-dienone |
| Molecular Weight (g/mol) | 243.29 |
| In vivo applications | Displays in vivo antitumor activity against human RXF 944XL renal xenografts in nude NMRI mice. Inhibits thioredoxin reductase with potencies correlated with their antiproliferative and cytotoxic efficacies. |
| In vitro applications | Potent activity against renal, colon, and breast cancer cell lines in the NCI 60-cell panel. Shows potent antitumor activity in HCT 116 and HT 29 human colon as well as MCF 7 and MDA 468 human breast cancer cell lines. |
| Research Area | Cancer |
| Storage | Ambient |
References: 6 entries
Chew et al. 2008. FASEB J. 22(6):2072-83. PMID: 18180330.
Thioredoxin reductase inhibition by antitumor quinols: a quinol pharmacophore effect correlating to antiproliferative activity.
Europe PMC ID: 18180330
Berry et al. 2005. J Med Chem. 48(2):639-44. PMID: 15658878.
Quinols as novel therapeutic agents. 2.(1) 4-(1-Arylsulfonylindol-2-yl)-4-hydroxycyclohexa-2,5-dien-1-ones and related agents as potent and selective antitumor agents.
Europe PMC ID: 15658878
Wells et al. 2003. J Med Chem. 46(4):532-41. PMID: 12570375.
4-Substituted 4-hydroxycyclohexa-2,5-dien-1-ones with selective activities against colon and renal cancer cell lines.
Europe PMC ID: 12570375
Add a reference
References: 6 entries
Chew et al. 2008. FASEB J. 22(6):2072-83. PMID: 18180330.
Thioredoxin reductase inhibition by antitumor quinols: a quinol pharmacophore effect correlating to antiproliferative activity.
Berry et al. 2005. J Med Chem. 48(2):639-44. PMID: 15658878.
Quinols as novel therapeutic agents. 2.(1) 4-(1-Arylsulfonylindol-2-yl)-4-hydroxycyclohexa-2,5-dien-1-ones and related agents as potent and selective antitumor agents.
Wells et al. 2003. J Med Chem. 46(4):532-41. PMID: 12570375.
4-Substituted 4-hydroxycyclohexa-2,5-dien-1-ones with selective activities against colon and renal cancer cell lines.
Add a reference