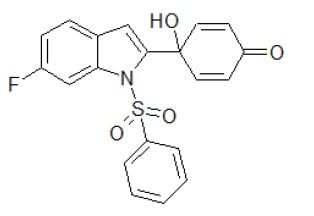
Antitumoral Quinol1h Small Molecule (Tool Compound)
Invented at University of Nottingham
- Datasheet
- References (6)
- Inventor Info
Info
| Catalogue Number | 151834 |
| Antigen/Gene or Protein Targets | Thioredoxin reductase |
| Type | Antitumoral |
| Relevance | A new class of molecule (4-hydroxy-4-hetarylcyclohexa-2,5-dien-1-ones) bearing a new pharmacophore (‘quinols’) has been identified with potent activity against renal, colon and breast cell lines in the NCI 60-cell panel. |
| Molecular Formula | C20H14FNO4S |
| lUPAC | 4-(1-Benzenesulfonyl-6-fluoro-1Hindol-2-yl)-4-hydroxy-cyclohexa-2,5-dienone |
| Molecular Weight (g/mol) | 383.40 |
| In vitro applications | Antitumor activity observed in HCT 116 (GI50 = 40 nM) and HT 29 (GI50 = 380 nM), and in breast cancer cell lines. Potent inhibitory effect in NCI 60 cells (GI50 =16 nM and LC50 = 2.24 µM). |
| Solubility | DMSO to 100 mM |
| Research Area | Cancer |
| Storage | -20C (desiccating conditions) |
| Notes |
Exhibits a mean GI50 value of 16 nM and a mean LC50 value of 2.24 uM in the NCI 60- cell-line screen, with LC50 activity in the HCT 116 human colon cancer cell line below 10 nM. Compound available for screening or similar studies. For larger quantities (>20mg) please contact Ximbio. |
References: 6 entries
Chew et al. 2008. FASEB J. 22(6):2072-83. PMID: 18180330.
Thioredoxin reductase inhibition by antitumor quinols: a quinol pharmacophore effect correlating to antiproliferative activity.
Europe PMC ID: 18180330
Berry et al. 2005. J Med Chem. 48(2):639-44. PMID: 15658878.
Quinols as novel therapeutic agents. 2.(1) 4-(1-Arylsulfonylindol-2-yl)-4-hydroxycyclohexa-2,5-dien-1-ones and related agents as potent and selective antitumor agents.
Europe PMC ID: 15658878
Wells et al. 2003. J Med Chem. 46(4):532-41. PMID: 12570375.
4-Substituted 4-hydroxycyclohexa-2,5-dien-1-ones with selective activities against colon and renal cancer cell lines.
Europe PMC ID: 12570375
Add a reference
References: 6 entries
Chew et al. 2008. FASEB J. 22(6):2072-83. PMID: 18180330.
Thioredoxin reductase inhibition by antitumor quinols: a quinol pharmacophore effect correlating to antiproliferative activity.
Berry et al. 2005. J Med Chem. 48(2):639-44. PMID: 15658878.
Quinols as novel therapeutic agents. 2.(1) 4-(1-Arylsulfonylindol-2-yl)-4-hydroxycyclohexa-2,5-dien-1-ones and related agents as potent and selective antitumor agents.
Wells et al. 2003. J Med Chem. 46(4):532-41. PMID: 12570375.
4-Substituted 4-hydroxycyclohexa-2,5-dien-1-ones with selective activities against colon and renal cancer cell lines.
Add a reference